Personalizing cancer care through precision medicine
Each of these initiatives will advance Markey’s commitment to precision medicine, an approach to cancer care that uses genetic analysis to help oncologists choose cancer therapies attuned to each patient’s individual needs.
“Oncology is now more genetic-based, whereas before it was based upon tumor types,” said Markey Director B. Mark Evers, MD, FACS. “By understanding the genetic makeup of our patients and their tumors, we can then help to direct their therapy.”
The Molecular Tumor Board launched in October 2016. It is co-directed by Markey’s Rachel W. Miller, MD, and Jill Kolesar, MS, PharmD, who recently joined Markey and the UK College of Pharmacy from the University of Wisconsin-Madison, where she started a similar initiative.
Evers hopes this wealth of information will make the Molecular Tumor Board a resource for cancer physicians across the Commonwealth in the future.
The Molecular Tumor Board will analyze the molecular characteristics of cancer tumors on a case-by-case basis and provide individualized recommendations for treatment. In doing so, it will also create a cache of information to direct the development of new therapies that target the types of cancers found at Markey and throughout Kentucky.
Evers hopes this wealth of information will make the Molecular Tumor Board a resource for cancer physicians across the Commonwealth in the future. Doctors would be able to send the molecular specifics of a patient’s tumor to the experts at Markey and receive advice on the latest treatment options that might work for that specific patient.
In addition to recommending therapies targeted to the specific genetic mutations that providers treat, the Molecular Tumor Board will also help drive the development of new therapies and clinical trials that specifically target these mutations, Miller said.
“The Molecular Tumor Board provides not only an opportunity for our patients, but also an opportunity for physicians to gain a better understanding of the function of the specific mutations and how to best target these molecular characteristics,” she said.
“Launching the center will help us bring more clinical trials to patients with cancer in Kentucky.”
– Jill Kolesar, MS, PharmD
To develop these new and novel therapies, Markey is launching the Early Phase Clinical Trials Center, which will focus on providing patients with increased access to phase I and phase II clinical trials. These early-phase studies can offer patients new treatment avenues for cancers that have proved difficult to target through standard therapies.
“Launching the center will help us bring more clinical trials to patients with cancer in Kentucky,” said Kolesar, who will serve as director of the center, drawing upon her experience in the clinical pharmacology of anti-cancer compounds and the implementation of clinical trials.
The center will encourage the development of phase I and II clinical trials at Markey – known as investigator-initiated trials – as well as seek out such trials at other institutions when appropriate.
Moreover, the center will have dedicated space and resources at Markey, ensuring that patients participating in early-phase trials will have the support they need. Patients will have access to navigators who will provide one-on-one support as well as to dedicated infusion nurses who will administer therapies and monitor patients. Working with this staff will be a multidisciplinary team of physicians, pharmacists and nurses.
“The center will provide patients with an enhanced experience,” Kolesar said. “We will ensure every patient receives individualized care and support, and they have the advantage of an interprofessional team working together to coordinate and provide their care.”
Evers said the two new endeavors represent a new phase in Markey’s commitment to precision medicine – one that will directly benefit patients across the Commonwealth.
“We’re using this as an opportunity to really reach out and personalize treatment for patients with cancer throughout Kentucky,” he said.
How the tumor board works
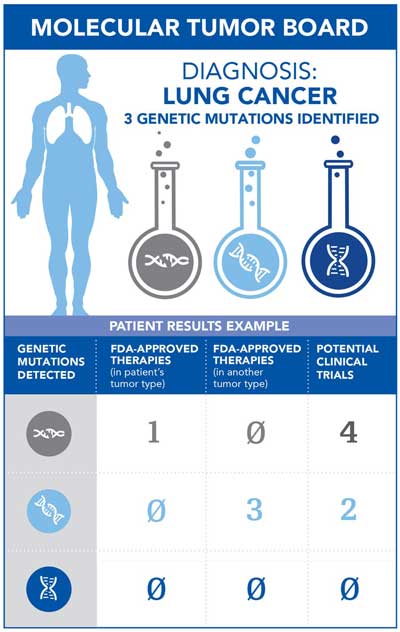 The patient is tested for all genetic mutations that are known to cause cancer, regardless of what type of tumor the patient has. In the example shown, a patient with lung cancer has three genetic mutations linked to cancer. The Molecular Tumor Board then uses these genetic mutations to consider possible treatment options for the patient. Each genetic mutation is evaluated for FDA-approved therapies for the patient’s tumor type, other FDA-approved therapies in other tumor types and any clinical trials available related to the specific genetic mutations. Taking into consideration the total treatment options available, the Molecular Tumor Board makes a treatment recommendation based on the best possible outcome for the patient.
The patient is tested for all genetic mutations that are known to cause cancer, regardless of what type of tumor the patient has. In the example shown, a patient with lung cancer has three genetic mutations linked to cancer. The Molecular Tumor Board then uses these genetic mutations to consider possible treatment options for the patient. Each genetic mutation is evaluated for FDA-approved therapies for the patient’s tumor type, other FDA-approved therapies in other tumor types and any clinical trials available related to the specific genetic mutations. Taking into consideration the total treatment options available, the Molecular Tumor Board makes a treatment recommendation based on the best possible outcome for the patient.

